

EUROLAB, with its state-of-the-art accredited laboratories and expert team, offers precise and rapid testing services within the scope of DIN 58953-1 test. This standard covers terms used in connection with the packaging of medical devices to be sterilized in final packaging.
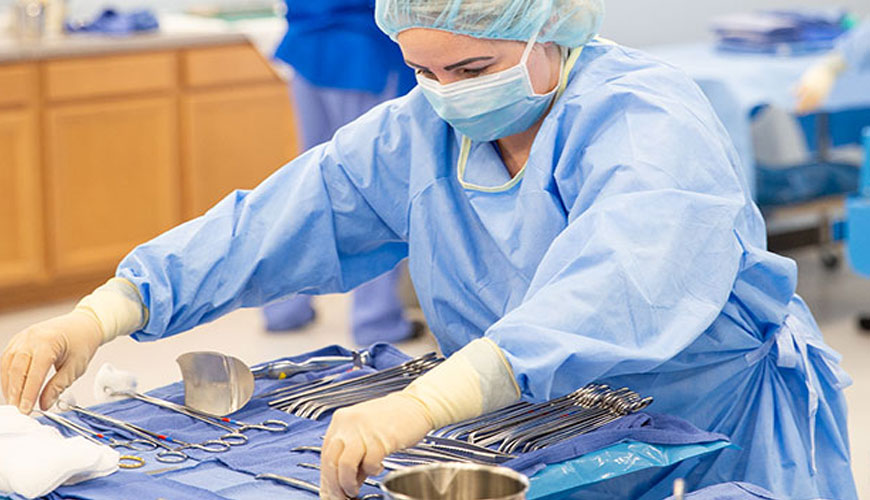
In order to reuse medical devices intended for low-germ or sterile use, cleaning, disinfection and sterilization processes are carried out after commissioning, including relevant work steps such as packaging and labeling.
All instruments, apparatus, devices, software, substances or other objects intended by the manufacturer specifically for diagnostic or therapeutic purposes and used individually or in combination, including software used by the manufacturer for the proper functioning of a human medical device, are intended to be used for the following purposes:
EUROLAB assists manufacturers with DIN 58953-1 test compliance. Our test experts, with their professional working mission and principles, provide you, our manufacturers and suppliers, the best service and controlled testing process in our laboratories. Thanks to these services, businesses receive more effective, high-performance and quality testing services and provide safe, fast and uninterrupted service to their customers.
To get an appointment, to get more detailed information or to request an evaluation, you can ask us to fill in our form and reach you.